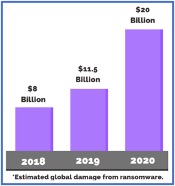
Two decades ago, California became the first state to legalize the medical use of cannabis. In 2021, medical use of cannabis is legal is 36 US states, and 17 states allow adult (‘recreational’) use. This trend of rapid legalization of the cannabis industry, while encouraging for industry growth, attracts more attention from federal regulatory bodies such as the Occupational Safety and Health Administration (OSHA). Following a number of incidents and near misses, cannabis facilities have been increasingly frequented by OSHA visits, leading to a spike in citations and fines. A review of past OSHA citations reveals that the most common citations in the cannabis industry pertains to the employer’s lack of awareness about the hazardous nature of some operations and materials handled in the facility. This leads to an absence of a formal fire prevention plan, lack of proper hazardous chemical training, deficiency in proper documentation related to workplace injury and limited evaluation of required personal protective equipment (PPE).1
Cannabis industry data suggests that as of today, an incident is often followed by an OSHA inspection. This naturally leads to the facility asking, ‘How do we prepare for an OSHA inspection and prevent future citations?’ The answer is a combination of identifying and mitigating risks in advance to avoid incidents and developing management systems that support the identification and risk mitigation efforts. Recent collaboration between cannabis business owners and organizations that write codes and standards have provided a framework in which to address the industry’s unique safety challenges to help reduce inherent risk to a facility. These codes and standards typically impact building construction/safety features and operation of the facility, however, additional risk mitigation can be drawn from the best practices already in place in process industries with similar hazards. These process industries have embraced process safety management (PSM) programs, which are built around principles flexible enough to be successfully implemented in the cannabis industry. Adopting such programs will serve the dual purpose of improving the overall safety record of the cannabis industry while enhancing company sustainability2 and help avoid events that lead to OSHA citations.
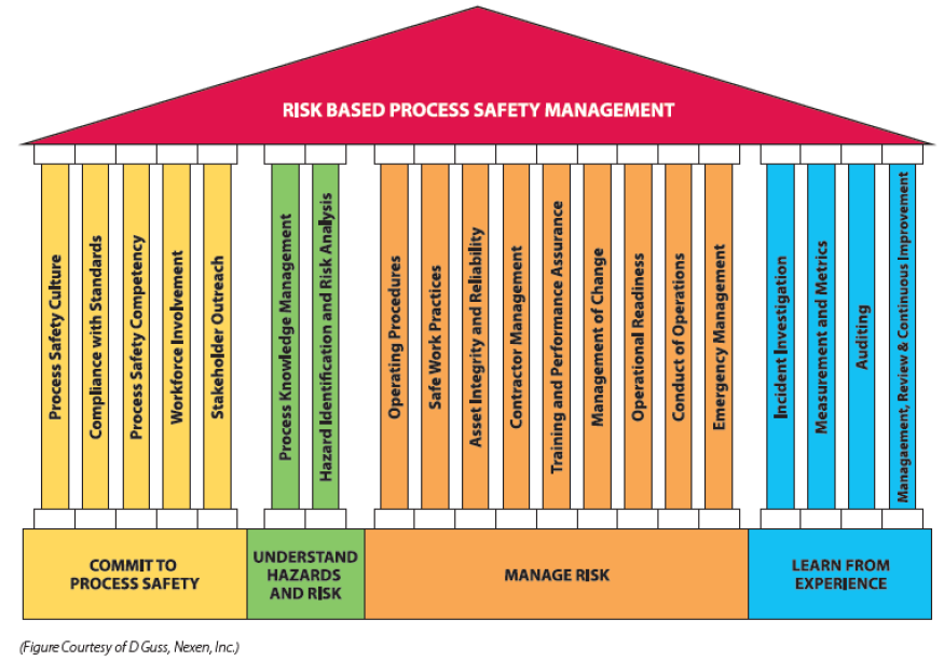
The risk-based process safety (RBPS) approach developed by the Center for Chemical Process Safety (CCPS)3 may prove to be the most effective framework to implement PSM programs in the cannabis industry. Unlike the prescriptive regulatory approach provided by OSHA 29 CFR 1910.119, the RBPS methodology recognizes that not all hazards and risks are equal. By assessing risk, an organization can develop an effective management system that will prioritize allocation of limited resources to address the highest risks. Figure 1 shows the four foundational blocks (pillars) of RBPS and the various elements that make up each pillar.
If a cannabis business owner were to develop programs on each of the pillars presented in Figure 1, a comprehensive safety program would be in place that delivers sustainable risk reduction and mitigation. However, as with any industry, the elements can be prioritized and tackled over time, starting with the elements having the most influence on the overall safety of a given facility. For example, a given facility may have great procedures and practices, but may not consistently train or instill employee knowledge or competency. Conversely, a facility may have personnel with great knowledge of hazards and risks, but are less developed with regard to documenting procedures, safe practices or training for new hires. Focusing available resources on the less developed elements will lead to an overall improvement in facility risk, leading to a lower likelihood of an incident and OSHA inspection.

As with any industry, positive and negative public perception is driven by the media, which tends to focus on attention-grabbing headlines. The majority of past incidents reported in the news for the cannabis industry were explosions that occurred during the extraction process. One such extraction explosion, shown in Figure 2, occurred in July 2015 at the New MexiCann Natural Medicine facility in Santa Fe, New Mexico. With a focus on the ‘hazard identification and risk analysis’ pillar of RBPS, future such events may be mitigated.
Of the twenty RBPS elements, hazard identification and risk analysis (HIRA) stands out as having the highest potential for immediate impact on the cannabis industry’s safety profile.
HIRA is a collection of activities carried out through the life cycle of a facility to ensure that the risks to employees and the public are constantly monitored to be within an organization’s risk tolerance. The four major areas to analyze are:
- Hazards – What are the possible deviations from the design intent?
- Consequences – What are the worst possible consequences (or severity) if any deviation occurs?
- Safeguards – Are there safeguards in the system to reduce the likelihood of this event?
- Risk – Is the risk within the tolerable level? If not, what steps are needed to reduce the risk? (Severity X Likelihood = Risk)
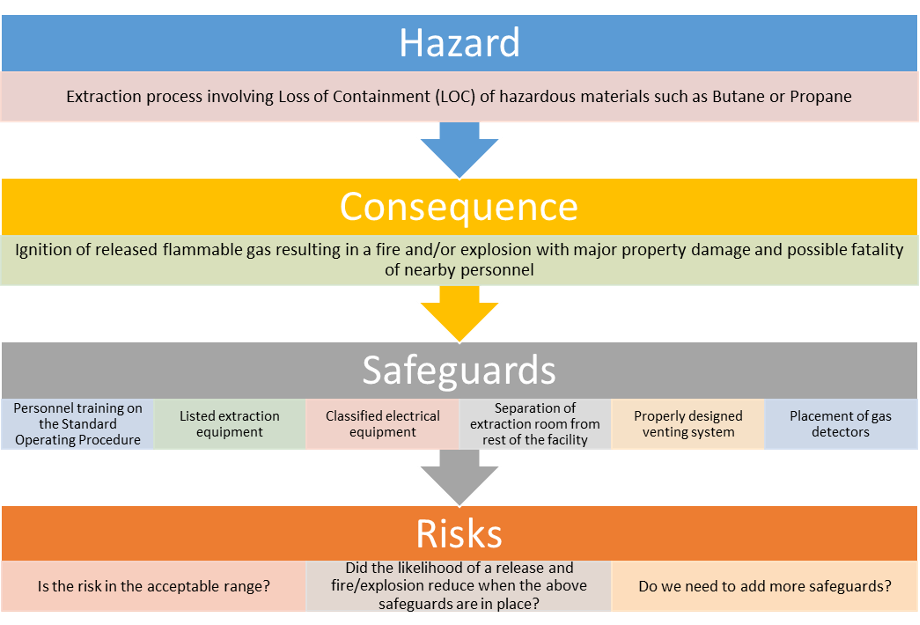
Let us consider an example case where the extraction process utilizes propane or butane as the extracting solvent. Figure 3 shows a simplified HIRA flow chart for the extraction process.
This systematic approach helps to understand the hazards and evaluate the associated risk. In addition, this approach highlights operator training as a crucial safeguard that can be credited to lower the overall risk of the extraction facility. Remember, lack of proper safety training (another element!) is one of the most cited OSHA violations in the cannabis industry. Another advantage to the HIRA methodology is that other safeguards that may be present can be identified, their effectiveness evaluated and additional risk reduction measures may be recognized. This will help business owners allocate their limited resources on the critical safeguards that provide the greatest risk reduction. Identifying, analyzing and solving for potential hazards is a key step in safe operation of a facility and avoiding OSHA citations.
While this article discusses only a single RBPS element, this example demonstrates how best practices from process industries can become a powerful tool for use in the cannabis industry. The “hazard identification and risk analysis” element of the RBPS approach is pertinent not only for the extraction process as discussed above, but also directly applicable to other aspects of the industry (e.g., dust explosions in harvesting and processing facilities, toxic impacts from fertilizers, hazards from the CO2 enrichment process in growing facilities, etc.).
References
- Top 5 OSHA Infractions for Cannabis Businesses
- The Business Case for Process Safety; 4th Edition; Center for Chemical Process Safety; 2018
- Guidelines for Risk Based Process Safety; Center for Chemical Process Safety: An AICHE Technology Alliance; published March 2007
- Video: Explosion rips through medical marijuana facility