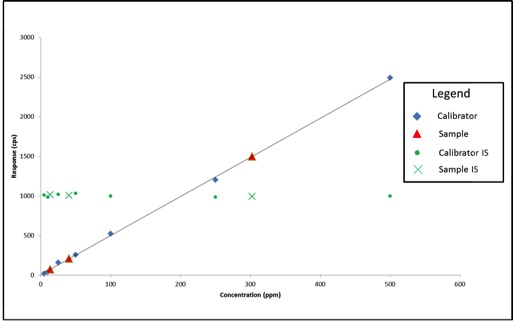
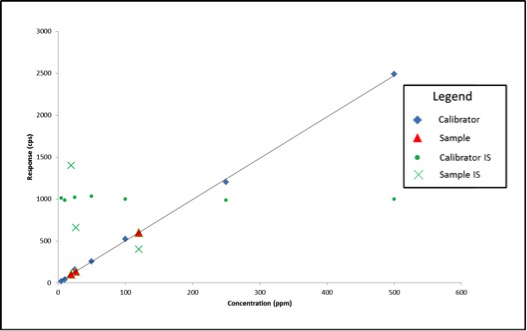
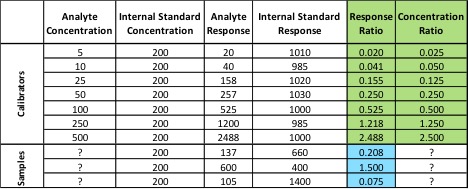
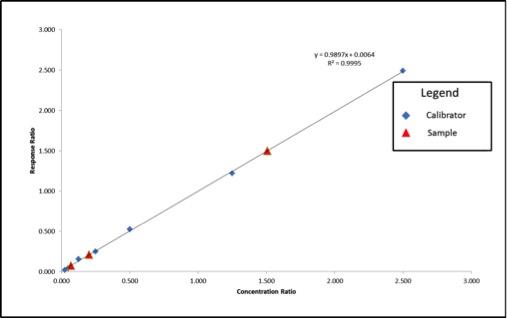
Last week, news of problems facing Oregon’s cannabis laboratory accreditation program surfaced, leading some to speculate about possible delays for the recreational cannabis market. According to The Register-Guard, ORELAP administrator Gary Ward believed the program was “on the precipice of collapse.”
 According to Jonathan Modie, spokesman for the Oregon Health Authority (OHA), the Oregon Environmental Laboratory Accreditation Program (ORELAP) was anticipating over 30 cannabis laboratories applying for accreditation and they doubled their staff from two to four to prepare for the uptick in applications.
According to Jonathan Modie, spokesman for the Oregon Health Authority (OHA), the Oregon Environmental Laboratory Accreditation Program (ORELAP) was anticipating over 30 cannabis laboratories applying for accreditation and they doubled their staff from two to four to prepare for the uptick in applications.
In June, the agency had zero labs applying for accreditation but within two months, 37 labs applied. However, the Oregon Department of Environmental Quality (DEQ) just provided three additional staff members on Monday to help with the application process, says Modie.
Some believe the issues could mean the state may not have enough accredited labs by October 1st, when the recreational cannabis market is expected to go into full swing. “It is difficult to say exactly how many labs we can accredit by October 1,” says Modie. “We have seven labs today which would bring it to nine labs waiting for assessment, but our goal is to get as many labs assessed and hopefully accredited as soon as possible.”
With the additional staff members, Modie is hopeful this will jumpstart the program. “We really appreciate our collaboration with the DEQ and look forward to boosting our capacity a bit to help us get through this busy time,” says Modie.
Part of the reason some laboratories might have trouble meeting prerequisites is simply because the requirements are very strict. “The process involves submitting a quality manual, standard operating procedures, method validation, submitting proficiency testing data and finally undergoing an ORELAP assessment by our staff, so it is a very rigorous process,” says Modie. “This speaks to our concern for making sure they have the right systems in place so public health is protected.” Modie said there were at least three labs that did not pass the assessment.

Bethany Sherman, chief executive officer of OG Analytical, believes the hardest part of the process involves getting accredited for testing pesticides. OG Analytical, based in Eugene, Oregon, has already received their accreditation, one of the first to do so. “The pesticide testing requires our most expensive instrumentation and the sample preparation for testing pesticides is the most time consuming,” says Sherman. “Not only does it require very specific instrumentation, it also requires a real know-how and expertise to ensure we are cleaning samples appropriately, minimizing background noise and looking at the pesticides in trace quantities.” According to Sherman, laboratories are also left to their own devices to develop methodologies specifically for the cannabis matrix, adding to the difficulties.
Rodger Voelker, Ph.D., lab director at OG Analytical, seems confident that the state will be able to handle it. “It is a relief they were able to get some resources from the DEQ and I think the state will not allow a program with this kind of importance to fall apart,” says Voelker. He believes after this initial phase of putting the program in place, the workload will go down. “It is easier to maintain a program than it is to implement,“ adds Voelker. In his eyes, it is crucial for the program to require rigorous science. “People are forced to reconcile that there is a tremendous amount of controls to be considered to produce legally defensible data and I think it is great that the requirements are so strict.”
The OHA’s job is to essentially safeguard public health and they do not want to leave any stone unturned when it comes to potential contamination, says Modie. “This is not just about getting as many labs accredited as possible, this is about protecting public health.”