During the COVID-19 pandemic, most testing laboratories have been classified as relevant for the system or as carrying out essential activities for national governments. Therefore, it is crucial to maintain activities and optimally assess the changes that are occurring, framed within the spread of the SARS-CoV-2 virus. Analytica Alimentaria GmbH, a testing laboratory with its headquarters in Berlin, Germany and a branch office in Almeria, Spain, decided to focus its management on the analysis of events and the options available, at the legal and employment level, to ensure continuity of activities and reducing, as much as possible, the damage for the parties involved: employees and company. Accredited by the International Accreditation Service (IAS) to ISO/IEC 17025:2017, Analytica Alimentaria GmbH is required to implement risk-based thinking to identify, assess and treat risks and opportunities for the laboratory. Since March 12, 2020 a crisis committee was established, formed by the six members of the company’s management, covering general management, human resources, direction of production, finance and IT. The committee meets every day and it intends to:
- Minimize the risks of contagion
- Be able to continue providing the service required by our clients

- ensure that the company as a whole will survive the economic impact of the crisis
- Take measures that are within the legality of both countries where the laboratory operates (Spain and Germany),
- Manage internal and external communication related to the crisis
To achieve correct decision making, daily meetings of the committee were established, to review the situations that were presented day after day and the actions that should be carried out. Each decision was analysed in a prioritized, objective, collaborative and global way.
The basis of the lab’s action plan was a well-developed risk assessment. In addition to the risk of getting a droplet or smear/contact infection with the coronavirus SARS-CoV-2 (risk I) by contact with other people, psychological stress caused by changing working conditions (home office), contact options and information channels were also identified (risk II).
As a result of the risk assessment, the conclusion was that a mix of various measures is the best form of prevention:
- Keep distance
- Avoid “super spreader” events
- Personal hygiene
- Regular communication between managers and personnel about the current situation and possible scenarios
The risk assessment took both areas into account. The following assessment was developed together with an external specialist and focused on risk I:
| Risk I |
Assessment |
Protective measures / hygiene plan |
| Organisation |
| Working hours and break arrangements |
High |
Limit the gathering of people and ensure a minimum distance:
- Relocated work, break and mealtimes
- Create fixed groups of shift-working staff
- Time gap of 20 min. between the shifts
- Enable home office wherever it is possible
|
| Third party access |
Moderate |
Few but “well-known” visitors:
- Reduce the number of visits and keep internal contacts to a minimum
- Ensure the contact chain
- Inform visitors about the internal rules and obtain written consent
|
| Dealing with
suspected cases |
High |
Isolation and immediate leave of the company:
- Contactless fever measurement (in case of typical symptoms)
- Leave the company or stay at home
- If the infection is confirmed, find contact persons (including customers or visitors) and inform them about a possible risk of infection
|
| Contact with other persons |
| Traffic route from home to work |
Moderate |
Avoid public transportation:
- Take a car, bicycle or go by foot
- Enable mobile work and teleworking
|
| At work |
High |
Always keep a sufficient distance of 2.0 m from people:
- If minimum distances cannot be maintained, wear protective masks or install physical barriers (acrylic glass)
- Organize traffic routes so that minimum distances can be maintained (one-way routes, floor markings indicating a distance of 2 m)
- Use digital meetings instead of physical ones
|
| Sanitary facilities |
Moderate |
Remove virus-loaded droplet as often as possible:
- Provide skin-friendly liquid soaps and towel dispensers
- Shorten or intensify cleaning intervals
- Hang out instructions for washing hands at the sink
- Include instructions for proper hand-disinfection
|
| Canteens, tea kitchens and break rooms |
High |
One person per 10 m² = minimum:
- Reduce the number of chairs per table
- Informative signs in every room, indicating the maximum number of permitted persons
|
| Ventilation |
High |
Diluting or removing bioaerosols (1 µm virus-droplets):
- Leave as many doors open as possible
- Regular and documented shock ventilation every 30 minutes or more frequently, depending on the size of window
- Operate ventilation and air-conditioning systems, since the transmission risk is classified as low here
|
| Use of work equipment |
Moderate |
Use tools and work equipment for personal use:
- Regular cleaning with changing use (PC, hand tools, coffee machine, …)
- If possible, use gloves when using equipment for a larger number of users
|
| Protective masks |
Moderate |
- Use of protective masks as an additional measure, indicating that this does not replace keeping distance
- Recommend wearing masks in commonly used areas and explain that they do not protect yourself, but help to protect others
- Give clear instructions (written and oral) on how to use a mask correctly and explain the use and purpose of different mask-types
- Distribute masks freely
|
A number of guidelines and concrete measures addressing the risks related to health issues are already in place. Those health issues in risk group II are more closely related to the psychological effects of the crisis, however, are also more complex to mitigate. The key strategy is communication and, in particular, actively listening to all employees of the company.
Analytica’s robust company culture, based on values established in coordination with the whole staff, has been of significant help during the crisis. The some 150 staff members are organized by over 22 team coordinators. During the crisis, active communication has been intensified significantly. The crisis management team set up regular alignment meetings with all the coordinators and with individual persons with particular situations. This way, not only was it possible to explain the development of the crisis and the subsequent measures, the conversations with coordinators were also the most important source of information enabling the appropriate decisions. The coordinators, closely aligned and in sync with management, were then able to communicate with their team members with a high degree of confidence. One outcome of the communication was a measure that proved very effective in fortifying trust within the company: all measures and evaluations, as well as a chronological review, are published in a dynamic internal report and are made available, with full transparency, to all staff members. Besides the many individual and group alignment meetings (usually held by video conference), this has been a key measure to establish confidence and security within the company.
 On the other hand, the company made a great effort to balance the effect of the general closure of kindergartens and schools in Spain and Germany. Each case where staff members were required to care for children at home was studied individually and agreements were established, adapting shifts and making use of time accounts, to allow childcare at home without significant loss of income.
On the other hand, the company made a great effort to balance the effect of the general closure of kindergartens and schools in Spain and Germany. Each case where staff members were required to care for children at home was studied individually and agreements were established, adapting shifts and making use of time accounts, to allow childcare at home without significant loss of income.
The success of the measures is shown by the continuous work of both laboratories during the crisis. Besides the personal tragedy of a possible infection, the identified risk to the company has the consequence of a (partial) quarantine due to an infected person in contact with the staff and the consequent loss of work-power which might lead, in extreme cases, to a closure of the laboratory. According to the governmental regulation in Germany, if an infection occurs (confirmed by the health department), contact persons cat. 1 (more than 15 min. contact face to face) are identified and sent to quarantine. Other contact persons, e.g. contact persons cat. 2 (same room without face to face) must be identified quickly with the collaboration of the infected person and notified and, if necessary, sent in quarantine. In this case, there is a confirmed emergency plan that maintains the laboratory’s ability to work, defining replacements and alternative work-flow strategies.
It has been part of our strategy to validate all our measures with the relevant guidance documents made available by the official competent institutions. The German Federal Office for Public Safety and Civil Protection (Bundesamt für Bevölkerungsschutz und Katastrophenhilfe) has published a guide, “Crisis Management in Companies, 9-point Checklist” especially for critical infrastructure companies in the CoVid-19 crisis.
Having been classified as a core business enterprise (Spain) and “relevant to the system” (Germany), we consider it important to use them as a reference to confirm our level of alignment with your proposal for crisis management.
An important effect, relevant to any leader in times of crisis, is that the confirmation of all points of such a checklist provides certain peace of mind regarding the question: Have we done everything we could?
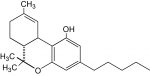
 The class is called Modern Cannabis Science and will involve a lot of genetic research. The course is sponsored by the Agricultural Genomics Foundation, a non-profit dedicated to scientific research and education in cannabis. In the press release, they describe the course as meant for students who are well informed, but “seek a deeper appreciation of scientific advancements in cannabis genetics.”
The class is called Modern Cannabis Science and will involve a lot of genetic research. The course is sponsored by the Agricultural Genomics Foundation, a non-profit dedicated to scientific research and education in cannabis. In the press release, they describe the course as meant for students who are well informed, but “seek a deeper appreciation of scientific advancements in cannabis genetics.”