As of this writing, one cannot register trademarks with the U.S. Patent and Trademark Office (USPTO) for cannabis products and services that “touch” the cannabis plant (i.e., cultivate, manufacture or dispense cannabis products), with the recent exception for certain hemp-based products and services, because use of trademarks must be lawful under federal law for federal trademark registration eligibility. Brand owners may, however, secure federal trademark registration protection for their brand names for certain cannabis-related products and services that are currently legal under federal law in advance of what could be the full legalization of cannabis at the state and federal levels.
Federal trademark registration provides brand owners with valuable benefits beyond common law (unregistered) and state registered trademark rights, including the preservation of national expansion rights and presumption of trademark ownership and validity. For those reasons, securing federal trademark registration protection for trademarks is a prudent business strategy.
 This article summarizes certain laws and regulations for securing federal trademark registration protection for cannabis products (including cannabidiol (CBD) products) and services. It also identifies other forms of intellectual property protection for cannabis businesses.
This article summarizes certain laws and regulations for securing federal trademark registration protection for cannabis products (including cannabidiol (CBD) products) and services. It also identifies other forms of intellectual property protection for cannabis businesses.
What Are Cannabis, Marijuana, Hemp and CBD?
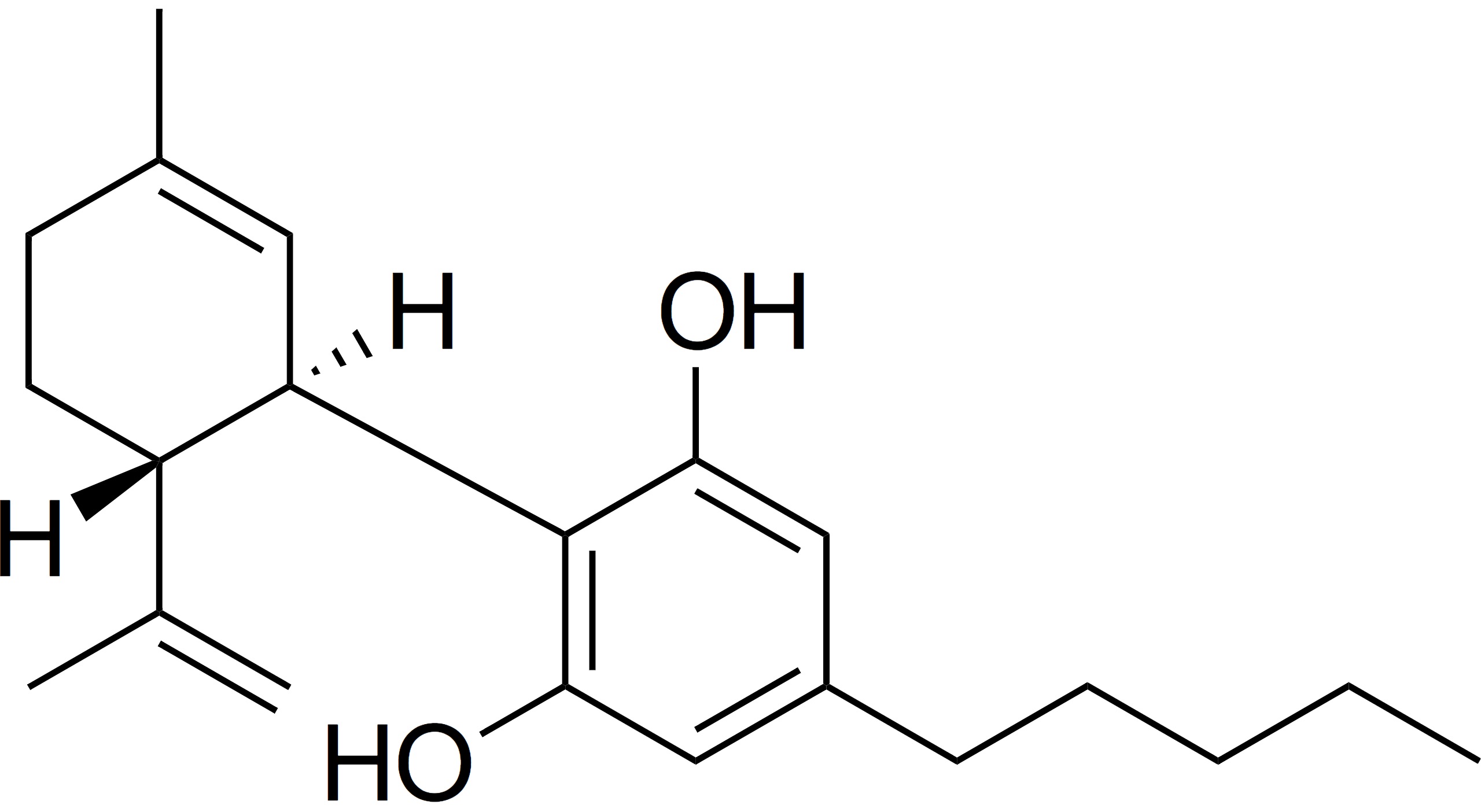
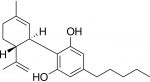
- Cannabis is a plant of the Cannabaceae family and contains many biologically active chemical compounds, including the well-known delta-9-tetrahydrocannabinol (THC) and cannabidiol (CBD) compounds.
- Parts of the Cannabis sativa plant are controlled under the Controlled Substances Act (CSA) under the drug class “marijuana.” The CSA is a federal law that regulates drug policy for the manufacture, importation, possession, use and distribution of certain substances. Marijuana is currently listed as an illegal Schedule I drug under the CSA, along with cocaine and heroin, due to its high potential for abuse, which is attributable mainly to the psychoactive effects of THC and the absence of a currently accepted medical use in the United States.
- Marijuana, a term the CSA uses, is the dried leaves of the cannabis plant. It is derived from the cannabis sativa and cannabis indica species and is used primarily as a psychoactive drug.
- Hemp is derived only from the cannabis sativa species and has historically been grown primarily for its strong fibers used for industrial purposes, including for making fabrics, clothing and rope.
- There is a significant difference between marijuana and hemp with respect to their concentration of THC, which gives the plant its psychoactive effect. While marijuana can reach THC levels of 30%, THC levels in hemp are typically 0.3% or less.
- The low level of THC in hemp is a reason why federal authorities recently removed it from the legal definition of marijuana, which means that cannabis plants and derivatives such as CBD derived from hemp that contain 0.3% or less of THC on a dry-weight basis are no longer considered controlled substances under the CSA.
- Cannabidiol (CBD) is an active ingredient in the cannabis plant and is derived primarily from the hemp plant. CBD has been touted for its many health benefits, including for the treatment of insomnia, pain and anxiety, and it has become a widely used ingredient in many types of products, including foods, cosmetics, building materials, industrial oils, plastics and textiles.
Relevant Laws and Regulations
Controlled Substances Act (CSA)
Under the CSA, the drug class marijuana is defined as “all parts of the plant Cannabis sativa L., whether growing or not; the seeds thereof; the resin extracted from any part of such plant; and every compound, manufacture, salt, derivative, mixture, or preparation of such plant, its seeds or resin” (subject to certain exceptions). 21 U.S.C. §802(16).
The CSA prohibits, among other things, manufacturing, distributing, dispensing or possessing cannabis that meets the definition of marijuana, including CBD derived from marijuana.
2018 Farm Bill Removes Hemp from the Definition of Marijuana
The 2018 Farm Bill signed into law on December 20, 2018, amended the Agricultural Marketing Act of 1946 and changed certain federal laws and regulations concerning the production and marketing of “hemp,” defined as “the plant Cannabis sativa L. and any part of that plant, including the seeds thereof and all derivatives, extracts, cannabinoids, isomers, acids, salts, and salts of isomers, whether growing or not, with a delta-9 tetrahydrocannabinol [THC] concentration of not more than 0.3 percent on a dry weight basis.”
- Those changes included removing hemp from the CSA’s definition of marijuana, which means that hemp and its derivatives, such as CBD derived from hemp, that contain no more than 0.3% THC on a dry-weight basis, are no longer controlled substances under the CSA.
- The recent change in the classification of hemp allows brand owners that legally manufacture and sell certain hemp-based products, including certain hemp-derived CBD products, to federally register their associated trademarks.
- However, the 2018 Farm Bill explicitly preserved FDA’s authority to regulate certain products containing cannabis or cannabis-derived compounds, even if derived from hemp, including CBD derived from hemp. Thus, federal laws, including FDA regulations, must still be considered for product legality before introducing products into commerce.
Food and Drug Administration (FDA)
Even with the removal of hemp from the CSA’s definition of marijuana, not all hemp-derived products are lawful following passage of the 2018 Farm Bill because certain products may still violate the Federal Food, Drug, and Cosmetic Act. For example, certain hemp-derived CBD products, including human foods, beverages, dietary supplements and animal foods, still violate FDA laws absent FDA approval.
The FDA monitors and investigates the sale of products that violate FDA laws, including CBD products promoted for therapeutic uses and treating diseases. When the FDA detects such violations, it may send warning letters to the violating parties as a first step in the enforcement process.
On December 20, 2018, the then FDA Commissioner Scott Gottlieb, M.D. made the following statement on that point:
“We’ll take enforcement action needed to protect public health against companies illegally selling cannabis and cannabis-derived products that can put consumers at risk and are being marketed in violation of the FDA’s authorities. The FDA has sent warning letters in the past to companies illegally selling CBD products that claimed to prevent, diagnose, treat, or cure serious diseases, such as cancer. Some of these products were in further violation of the FD&C Act because they were marketed as dietary supplements or because they involved the addition of CBD to food.”
Furthermore, in a recent letter to a company selling CBD products, the FTC sent a joint letter with the FDA, and that letter included the following statements and warnings:
-
“The FTC strongly urges you to review all claims for your products and ensure that those claims are supported by competent and reliable scientific evidence. Violations of the FTC Act may result in legal action seeking a Federal District Court injunction or Administrative Cease and Desist Order. An order also may require that you pay back money to consumers.
-
You should take prompt action to correct the violations cited in this letter. Failure to promptly correct violations may result in legal action without further notice, including, without limitation, seizure and/or injunction.”
What about using hulled hemp seed, hemp seed protein powder and hemp seed oil in human food?
- In December 2018, the FDA generally recognized as safe (GRAS) hulled hemp seed, hemp seed protein powder and hemp seed oil. Accordingly, the FDA’s current position suggests that those products may legally be marketed in human foods for the uses described in the notices, provided they comply with all other requirements. To date, the FDA has not received any GRAS notices for the use of hemp-derived ingredients in animal food.
- Hemp seeds are the seeds of the Cannabis sativa plant. They do not naturally contain THC or CBD. The hemp seed-derived ingredients that are the subjects of the GRAS notices contain only trace amounts of CBD and THC. The FDA has reported that “[c]onsumption of these hemp seed-derived ingredients is not capable of making consumers ‘high.’”
- Those GRAS conclusions do not affect the FDA’s position on the addition of CBD and THC to food.
U.S. Trademark Registration Eligibility
Trademarks Must Be Used for Lawful Activities
A trademark’s use must be lawful under federal law for federal trademark registration eligibility. Whether activities associated with cannabis and/or cannabis-related goods or services are lawful under federal law requires review of various federal laws, including the Federal Food, Drug, and Cosmetic Act.
Federal law controls federal trademark registration eligibility, period.
If a trademark application is filed for goods or services that violate federal laws, including for marijuana products and/or services or certain products that feature CBD, such as foods and nutritional supplements, the USPTO Examiner should refuse the application. Furthermore, filing an “intent-to-use” trademark application cannot obviate that refusal.
What does that mean? It means that filing a trademark application based on an “intent to use” the trademark “in the future” in anticipation of federal law legalizing cannabis still violates current law (the law as of the application filing date), and thus the application should be rejected because the applicant does not and cannot have a “bona fide intent” to use the applied-for mark for a legal purpose.
The USPTO Examination Guide 1-19 for examining cannabis marks states that:
“[r]egistration of marks for foods, beverages, dietary supplements, or pet treats containing CBD will still be refused as unlawful under the FDCA, even if derived from hemp, as such goods may not be introduced lawfully into interstate commerce.”
The following is an excerpt from an issued Trademark Office action refusing registration of a mark on the basis the listed cannabis goods are unlawful:
“Registration is refused because applicant does not have a bona fide intent to lawfully use the applied-for mark in commerce.
To qualify for federal trademark/service mark registration, the use of a mark in commerce must be lawful. Gray v. Daffy Dan’s Bargaintown, 823 F.2d 522, 526, 3 USPQ2d 1306, 1308 (Fed. Cir. 1987) (stating that “[a] valid application cannot be filed at all for registration of a mark without ‘lawful use in commerce’”); TMEP §907; see In re Stellar Int’l, Inc., 159 USPQ 48, 50-51 (TTAB 1968); Coahoma Chemical Co., Inc. v. Smith, 113 USPQ 413 (Com’r Pat. & Trademarks 1957) (concluding that “use of a mark in connection with unlawful shipments in interstate commerce is not use of a mark in commerce which the [Office] may recognize.”). Thus, the goods and/or services to which the mark is applied must comply with all applicable federal laws. See In re Brown, 119 USPQ2d 1350, 1351 (TTAB 2016) (citing In re Midwest Tennis & Track Co., 29 USPQ2d 1386, 1386 n.2 (TTAB 1993) (noting that “[i]t is settled that the Trademark Act’s requirement of ‘use in commerce,’ means a ‘lawful use in commerce’”)); In re Pepcom Indus., Inc., 192 USPQ 400, 401 (TTAB 1976); TMEP §907.
Here, the items or activities to which the proposed mark will be applied are unlawful under the federal Controlled Substances Act (CSA), 21 U.S.C. §§801-971.”
USPTO Guidelines for Marijuana and Hemp Products: Key Takeaways
- Trademark registrations for marijuana and marijuana by-products, including CBD derived from marijuana, are still unavailable.
- Trademark registrations for certain hemp products are available. If an applicant’s goods are derived from hemp, as defined in the 2018 Farm Bill, the identification of goods must specify that they are derived from hemp and that the products contain less than 0.3% THC. Thus, the scope of the resulting registration will be limited to goods compliant with federal law.
- Trademark applications covering certain CBD infused products, including foods, beverages, dietary supplements and pet foods, are still refused, even if derived from hemp, because such goods may not be introduced lawfully into commerce without FDA approval.
- The USPTO is currently approving trademarks for skin care preparations and cosmetics that feature hemp ingredients, including CBD derived from hemp, as long as the application complies with the 2018 Farm Bill and USPTO filing requirements.
- If a pending application’s filing date is prior to December 20, 2018 (the effective date of the 2018 Farm Bill), the applicant must amend the filing date to a date later than December 20, 2018 before the application may proceed. Once the date has been amended, a new search is conducted for any prior pending confusingly similar marks.
- Trademark applications for hemp cultivation and production, if allowed, will require proof of authorization and licensure in accordance with a plan approved by the U.S. Department of Agriculture.
Federal Trademark Registration Considerations and Options
Although marijuana products and services (i.e., products and services that “touch the plant”) and certain hemp-based products are currently illegal under federal law, making their associated marks ineligible for federal trademark registration protection, there are still certain cannabis-related activities that are legal and thus eligible for federal trademark registration.
Examples of legal activities include:
- Providing informational services related to cannabis or marijuana-related goods and services.
- Clothing, including t-shirts and hats, featuring a cannabis-related trademark.
- Educational programs in the fields of cannabis and CBD, including for health benefits and therapeutic uses of medical cannabis and CBD.
- Providing an internet news portal featuring links to current events, information, commentary, non-downloadable publications in the nature of brochures, articles, and non-downloadable multimedia files containing video, audio or text in the fields of cannabis or cannabis news.
- Online journals, namely blogs featuring information about cannabis.
- Entertainment services, namely, providing podcasts featuring medical and industry experts in the field of cannabis and medical marijuana.
If a brand owner secures federal trademark registration protection for marks for legal activities, including those listed above, those trademark registrations and rights may arguably preserve future product and service expansion under the same registered mark for “related” goods and/or services that are unlawful as of the trademark application filing date, but later become lawful, including CBD infused foods and nutritional supplements and marijuana itself.
Why? Because trademark law protects consumers from “source confusion.”
- For example, if a brand owner adopts the trademark N-DuraRun for running shoes, another party may not adopt the same or confusingly similar mark for running pants because consumers would likely be confused as to the source of running shoes and running pants if offered under the same trademark by different parties.
- It is not confusion as to what a consumer is buying (“I thought I was buying running shoes… instead I mistakenly purchased running pants…”). Rather, it is confusion as to the source of the products (“I purchased EnDuraRun brand running pants because I thought they were made by the same company that makes N-DuraRun brand running shoes!”).
- A question to ask is “Would the average consumer reasonably believe that the parties’ respective goods are of the type that would originate from the same source?”
- If the answer is “yes” and if the parties’ respective marks are confusingly similar, there may be a likelihood of consumer confusion as to the source of the parties’ respective goods.
For example, if a company provides informational services in the field of cannabis and cannabis derivatives, including CBD infused foods, and/or provides foods and nutritional supplements featuring hemp seed protein powder and hemp seed oil, and it secures federal trademark registration protection for its trademark for those goods and/or services, that existing federal trademark registration and rights may arguably preserve the brand owner’s right to use and register the same mark for “related” goods and services, which could include CBD-infused foods and nutritional supplements if/when those goods become legal. That is so because the average consumer would arguably believe that informational services about CBD infused foods and CBD infused foods themselves would originate from the same source and also believe that foods and nutritional supplements featuring hemp seed protein powder and hemp seed oil and foods and nutritional supplements featuring hemp-derived CBD would originate from the same source.
Source confusion is the crux of trademark law.
Therefore, securing federal trademark registration protection now for goods and services that are lawful can preserve future trademark rights for cannabis-related products and services that are currently unlawful and may avoid losing valuable trademark rights to third parties.
As companies prepare for the potential federal legalization of all forms of cannabis, securing federal trademark registration now for brand names for goods and services that are currently legal is vital for protecting valuable company assets, current and future business opportunities, and future growth, and it is possible as long as brand owners understand the current status of the regulatory landscape and the intricacies of trademark law.
Other Forms of Intellectual Property Protection
In addition to trademark and federal trademark registration protection, there are other intellectual property protections available for marijuana, hemp and cannabis businesses, including:
- State trademark filings. In states that have legalized cannabis, state trademark registrations may be available.
- Common law trademark rights. In states that have legalized cannabis, common law trademark rights may be available.
- Patent protection. Patent protection may be secured for various inventions, including plants, such as new strains of the cannabis plant, and methods of cannabis hydration and lighting.
- Trade secrets. Trade secrets can protect certain aspects of a business, including formulas, processes or methods, that are not generally known or reasonably ascertainable by others and that can help a business obtain an economic advantage over competitors or customers. To be eligible as trade secrets, however, a business owner must take the necessary steps to legally protect them or they will be lost.
- Copyrights. Copyright protection may be secured for certain company creative works, including trademark logos (artwork), written materials, photographs and software.
As the laws governing the cannabis industry continue to evolve, including trademark, FDA and banking laws and regulations, all interested parties, including cannabis business owners, law firms and investors, must stay abreast of the rapidly changing legal landscape to maximize business growth opportunities, ensure proper legal and regulatory compliance, and avoid having their businesses go up in smoke.
Notice: This article is for educational purposes only, is not legal advice and should not be substituted for retaining an attorney.














 Corby Iannuzzelli: Our product is completely digital. The consumer experience starts with integration at the online checkout. When it’s an e-commerce shopping cart and somebody is placing an order, they will see a button called “Pay with KindTap.” The first time they click that button they’re automatically brought to our integrated web app where they do a quick and easy application for our digital revolving line of credit product. If approved, they instantly go back to the checkout screen and their first purchase will just happen immediately, with flexible payment options over time. If the consumer decides they don’t want our KindTap credit and would rather have a pay now-product where we pull the funds from their bank account, then the consumer can do so. So, there is no physical card per se, it’s integrated like PayPal or Affirm at the point of checkout online. For the consumers who use KindTap credit, there is a mobile app where they can see their transactions, view statements, pay their bills, etc.
Corby Iannuzzelli: Our product is completely digital. The consumer experience starts with integration at the online checkout. When it’s an e-commerce shopping cart and somebody is placing an order, they will see a button called “Pay with KindTap.” The first time they click that button they’re automatically brought to our integrated web app where they do a quick and easy application for our digital revolving line of credit product. If approved, they instantly go back to the checkout screen and their first purchase will just happen immediately, with flexible payment options over time. If the consumer decides they don’t want our KindTap credit and would rather have a pay now-product where we pull the funds from their bank account, then the consumer can do so. So, there is no physical card per se, it’s integrated like PayPal or Affirm at the point of checkout online. For the consumers who use KindTap credit, there is a mobile app where they can see their transactions, view statements, pay their bills, etc.