Part 1 of this series discussed the lack of bankruptcy protections for cannabis companies, since bankruptcy in the U.S. is an exclusively federal procedure and cannabis remains illegal under federal law and proposed a number of alternative options for businesses struggling in the current environment. Part 2 of this series focuses on one of these alternatives: state law receiverships.
Background
A cannabis operation facing financial difficulties may try to avail itself, on the one hand, of the contractual remedies described in Part 1 of this series, but these remedies may be flimsy given their narrow scope and reliance on voluntary negotiation between parties whose relationship is already likely tense; on the other hand, the statutory remedies described in Part 1 of this series may be too rigid and absolute, necessitating the disposition of a business as a collection of assets, instead of its continued operation as a going concern. An alternative is receivership, a flexible but powerful quasi-judicial approach paralleling federal bankruptcy able to be administered by state courts. Compared to federal bankruptcy, state receivership is both over and under-inclusive: while receivership can be used in many more situations than insolvency, such as a financially healthy business that is nonetheless subject to regulatory action, receivership provides less comprehensive protection for an insolvent business.
Receiverships have their roots in English and Welsh courts of equity, which were seen as offering fairer remedies than their contemporary common law courts, bound as they were by ponderous precedent. In contrast, courts of equity had more discretion to apply remedies which could be more tailored and “equitable” to an individual petitioner, even if such remedies were not codified. While this separation of equitable and common law courts does not generally exist in the modern U.S. legal system (except for a few hold-out states, most notably, Delaware), the legacy remains in the type of civil remedy available: while most remedies are awarded as monetary redress for a past wrong suffered by a plaintiff (e.g. liquidated damages for the discloser of confidential information or the “benefit of the bargain” for the seller of a company), equitable remedies often require prospective action (or forbearance of an action) by the defendant (e.g. an injunction on disclosure by a recipient of confidential information or specific performance by a purchaser of a company). To draw the analogy out, bankruptcy is a “legal” process to address insolvency since it is governed by a comprehensive regime of federal statutes and rules in the Bankruptcy Code (which is, ironically, applied by specialized federal courts), while receivership is the “equitable” side of the same coin: a judicially-created remedy to manage or liquidate a business, among other actions, where it would not be equitable (or, most importantly for cannabis businesses, not possible) for a bankruptcy action.
Some states with legalized cannabis have cannabis-specific receivership statutes, usually providing that the receiver either be temporarily or fully licensed similar to any other operator of a cannabis business.As an equitable remedy used by various states and federal entities, generalizations about the receivership process are difficult to make. However, broadly speaking, a typical receivership process begins with a complaint filed against the entity for which receivership is sought in state court. This filing can be made by a variety of parties outside of the standard debtor-creditor relationship (reflecting the equitable nature of receiverships), including by regulators and disputing owners of a business. After this filing, a motion to appoint the receiver (which is usually but not always a third party) is filed with the court; consent of the opposing party is generally not required in appointing a receiver but can often make the process easier. The complainant must then establish standing and the occurrence of certain events, including insolvency, but also mismanagement of a corporation or a foreclosure. The requirements of such events are fact-specific and may often be governed by statute or the contractual relationship between parties. The order appointing the receiver usually sets out the specific powers the receiver has in any given case to oversee the disposition or operation of the assets subject to the receivership (called the “receivership estate”) for the benefit of its’s creditors.
Receivership laws generally fall into two categories: some states provide for a broad general statute, sometimes accompanied by statutes specific to industries which are heavily regulated, entity types, or process, while in other states the power is an extension of the court’s powers, set forth in the state’s rules of civil procedure. States also differ as to whether a receivership is considered an independent remedy, a standalone legal action which can be pursued in and of itself (e.g. a petition by a creditor to appoint a receiver to resolve settle an unpaid debt), or an ancillary remedy, a legal action that supports a primary claim (e.g. a request to appoint a receiver in connection with a dispute over the ownership of a business). Some states provide for general receiverships, which allows receivers to take control of an entire business, while other states also allow limited receiverships, which allows the receiver take control of a portion of a business, while the owner operates the remainder. Some states with legalized cannabis have cannabis-specific receivership statutes, usually providing that the receiver either be temporarily or fully licensed similar to any other operator of a cannabis business.1
Below is an overview of the laws and rules governing receiverships in certain states which have legalized cannabis.
Arizona
 In Arizona, receivership is governed by statute, with a general statute and specific statutes for certain industries and type of receivership. Arizona law recognizes that principles of equity apply to all matters relating to receivers, providing the court overseeing the receivership with additional power to decide the remedies available to the receiver. In addition, Arizona has enacted a specific statutory framework for the appointment of receivers for commercial real property and personal property related to or used in operating the real property. Arizona also uses a separate receivership statute to provide for corporate dissolution receiverships, in which a court in a judicial corporate dissolution proceeding may appoint one or more receivers to wind-up, liquidate, or manage the business and affairs of the corporation.
In Arizona, receivership is governed by statute, with a general statute and specific statutes for certain industries and type of receivership. Arizona law recognizes that principles of equity apply to all matters relating to receivers, providing the court overseeing the receivership with additional power to decide the remedies available to the receiver. In addition, Arizona has enacted a specific statutory framework for the appointment of receivers for commercial real property and personal property related to or used in operating the real property. Arizona also uses a separate receivership statute to provide for corporate dissolution receiverships, in which a court in a judicial corporate dissolution proceeding may appoint one or more receivers to wind-up, liquidate, or manage the business and affairs of the corporation.
There are no specific statutes governing receiverships of cannabis businesses, so the general receivership statute applies to cannabis businesses, subject to Arizona’s rules governing the operation of a cannabis business. For example, Arizona cannabis regulations that require anyone volunteering or working at a medical or recreational cannabis dispensary to be registered with the cannabis regulator similarly apply to a receiver appointed over a licensed cannabis business.
California
California does not have significant entity-specific or industry-specific statutes for receiverships; rather a California court’s power to appoint a receiver is granted under the state’s rules of civil procedure. Receiverships in California are solely an ancillary remedy; a receivership is commenced once a complaint is filed and any party to the action may seek to appoint a receiver. Circumstances that allow for the appointment of a receiver are fact-specific and at the discretion of a judge, although contractual provisions for the appointment of a receiver are given weight under the rules. Sales of assets in the receivership estate must be submitted to, and approved by, the appointing court.
 While the rules of civil procedure provide for the general powers of a receiver, the specific powers a receiver possesses in any given case is granted by the judicial order appointing the receiver; this appointment order is therefore, along with the court itself, the primary authority for the parties in any given receivership. California explicitly disqualifies certain persons, such as parties to the lawsuit, an attorney of a party, a person interested in an action, or any person related to any judge of the court within the third degree, as receivers.
While the rules of civil procedure provide for the general powers of a receiver, the specific powers a receiver possesses in any given case is granted by the judicial order appointing the receiver; this appointment order is therefore, along with the court itself, the primary authority for the parties in any given receivership. California explicitly disqualifies certain persons, such as parties to the lawsuit, an attorney of a party, a person interested in an action, or any person related to any judge of the court within the third degree, as receivers.
While California’s receivership rules do not explicitly contemplate cannabis businesses, receiverships for cannabis companies have taken place, but in our experience are less common in California than assignments for the benefit of creditors (which we will address in a later article). Like other licensed businesses in California, cannabis companies must provide notice to the state regulatory agency which granted the license. It is up to the agency’s discretion whether the business may be operated under the existing license or whether the receiver must secure a new or temporary license.
Colorado
Like California, no generally applicable receivership statute exists in Colorado; instead, receiverships are governed by the state’s rules of civil procedure. Under these rules, a receiver can be appointed under a court’s general equitable powers. Appointment of a receiver is an independent remedy in Colorado, but is contingent on a lawsuit having commenced and the court having deemed the receivership as necessary and proper. In addition to the court’s general equitable powers to appoint a receiver, and unlike California, Colorado has receivership statutes that are entity and industry specific. The entity-specific statutes permit the appointment of a receiver for the judicial dissolution of for-profit corporations, non-profit corporations, limited liability companies, and cooperatives, and the industry-specific statutes permit the appointment of a receiver for the windup of failed insurance companies and the closure of long-term care facilities.
Similar to California, the court order appointing a receiver governs the entire receivership process and any disposition of the assets of the receivership estate must be submitted to and approved by the court.
 As befitting the first state to legalize adult-use cannabis, Colorado’s cannabis regulations specifically address receiverships: the rules create a notice and application requirement for all court appointees, including receiverships, and require receivers to register with the regulator as a “temporary appointee” of the court.
As befitting the first state to legalize adult-use cannabis, Colorado’s cannabis regulations specifically address receiverships: the rules create a notice and application requirement for all court appointees, including receiverships, and require receivers to register with the regulator as a “temporary appointee” of the court.
Illinois
Illinois does not have a comprehensive receivership statute; instead, the state has industry-specific statutes, including for regulated industries such as nursing home facilities and telecommunication carriers. Illinois also provides for “equity receiverships”, which are used as an ancillary remedy in business disputes in order to stabilize a business that is adversely affected by fraud, neglect, waste, dissipation, or other misconduct during the pendency of the underlying proceeding. If the underlying matter is within the general or statutory jurisdiction of the court, then such court has jurisdiction over the receivership.
There are no specific statutes governing receiverships of cannabis businesses, but the governing statute does contemplate operation of a cannabis business by a receiver, so regulations promulgated thereunder should apply to receivers as well, including with respect to licensing.
Maryland
Adult-use cannabis sales only began in Maryland July 1, 2023. Maryland has a general receivership statute.
Receivers in Maryland are generally appointed by the person seeking appointment, including the court, and must meet certain qualifications, such as not having any material financial interest in the outcome of the receivership, and not having any debtor-creditor relationship with or equity interest in any party to the receivership. While the general receivership statute provides for broad powers of the receiver, including general management of receivership property, hiring professionals, and issuing subpoenas, the court may modify or expand the powers of the receiver via the appointment order.
 While there is no cannabis-specific receivership statute, Maryland’s medical cannabis rules contemplate and authorize the transfer of licenses to a receivership; similar rules have been proposed for adult-use cannabis licenses as well.
While there is no cannabis-specific receivership statute, Maryland’s medical cannabis rules contemplate and authorize the transfer of licenses to a receivership; similar rules have been proposed for adult-use cannabis licenses as well.
Nevada
Nevada has a broad receivership statute, in addition to both entity and industry specific statutes. Case law is not well-developed and mostly predates the current statutory scheme, but there is support for a receiver being appointed outside of a statutory context, specifically when the situation is governed by contractual agreement.
The general receivership statute provides that a receiver may be appointed in a variety of situations, such as fraudulent property purchases, foreclosure of mortgages, or the dissolution or insolvency of a corporation.
Nevada has a statutory regime for receiverships for cannabis companies. Unlike the general statute, there are significant requirements for who can be a receiver for a cannabis business. A receiver must first secure a cannabis establishment agent registration card for a cannabis receiver issued by Nevada’s cannabis regulator. In addition, the receiver must submit an application to the regulator accompanied by, among other requirements, a statement saying the receiver has not previously had an agent registration card revoked. The receiver must also provide proof that she has (1) experience or knowledge of the cannabis industry, (2) experience as a receiver appointed by a court, (3) knowledge and skills necessary to make reasonable financial decisions, and (4) adequate financial capacity to fulfill the duties of a receiver. If the regulator is satisfied with the receiver’s application, it will issue the receiver an agent registration card which must be renewed two years after issuance. It is worth noting that Nevada’s statute governing the non-transferability of certain agent registration cards for cannabis allows the regulator to adopt regulations that give priority in the processing of transfers of licenses for transferors subject to receivership. To date, however, no such regulations allowing priority for receivership processing have been adopted.
Washington
Washington has a general receivership statute, but not any entity or industry-specific receivership statutes. Washington’s receivership structure with overhauled in 2004 with the passage of a new law, so it is not completely settled whether receivership is now an independent or ancillary remedy; however, the language of the statute language suggests that it is an independent remedy.
To be appointed a receiver in Washington, the individual must meet certain requirements, including not being a party to, or be closely controlled by a party to, the underlying action and not having materially adverse interest to the person against whom receivership is sought. The general statute specifically outlines the powers of the receiver. Certain actions by the receiver require court approval before being finalized, including the assumption or rejection of executory contracts, and sales of property outside the ordinary course of business.
Washington law specifically provides for receiverships for cannabis companies. To be a receiver, the person must satisfy the requirements of Washington’s receivership law, and either be preapproved by the cannabis regulator or else be approved post-application. In order to qualify for the regulator’s preapproved receiver list, or be approved post-application, the putative receiver must (1) submit an application, (2) have been a Washington resident for at least six months prior to submission, (3) submit to and pass a criminal background check, (4) provide financial disclosures as requested by the regulator, and (5) disclose any interests in the cannabis licensees. Once a person is appointed as receiver for a cannabis licensee, she shall not have a financial interest in, or simultaneously serve as receiver for, another licensed cannabis retailer. The receiver may not also serve as a receiver for, or be a party of interest in, more than five cannabis retail licensees or more than three cannabis producer and/or processor licensees at the same time. Finally, any person who files a receivership action involving a cannabis licensee must provide notice to the regulator.
Part 3 of this series on Alternatives to Bankruptcy for Cannabis Companies continue our review of receivership in various states and other bankruptcy alternatives, including assignments for the benefit of creditors.
Reference
- As cannabis legalization continues to spread, more robust industry-specific receivership rules may be promising given the heavily regulated and specialized nature of the business, similar to how a number of states have industry-specific rules for other heavily regulated industries.
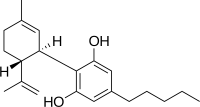
 Safety is indeed a complex subject, especially when it comes to cannabis products. For instance, acceptable limits for microbial contaminants for the inflorescence of a Cannabis sativa L. plant are quite literally all over the map. What about the route of administration? Take an inhaled product (like a vape pen) vs. an ingested product (e.g. an edible). Many outdoorsy people like myself may enjoy the smell of cooking s’mores over a campfire, but the particles and VOCs can irritate our lungs, especially when exposed over long periods of time. We aren’t inhaling those s’mores – so the production of the marshmallow, chocolate and graham crackers come with different risks we need to evaluate. It’s not just about the product – it’s how it’s consumed.
Safety is indeed a complex subject, especially when it comes to cannabis products. For instance, acceptable limits for microbial contaminants for the inflorescence of a Cannabis sativa L. plant are quite literally all over the map. What about the route of administration? Take an inhaled product (like a vape pen) vs. an ingested product (e.g. an edible). Many outdoorsy people like myself may enjoy the smell of cooking s’mores over a campfire, but the particles and VOCs can irritate our lungs, especially when exposed over long periods of time. We aren’t inhaling those s’mores – so the production of the marshmallow, chocolate and graham crackers come with different risks we need to evaluate. It’s not just about the product – it’s how it’s consumed. At the FDA, ASTM standards are used every day to keep us safe. Whether it is to measure the absorbency of tampons (21 CFR 801.430), enforce safety specifications of synthetic and natural wax coatings that are used to coat much of our produce, gummies and more (21 CFR 178.3770), quantifying the impurities in our bottled water (21 CFR 165.110) and many more.They have long been recognized as the de facto minimum standards that balance the need to protect consumers without imposing undue burdens on innovation by industry.
At the FDA, ASTM standards are used every day to keep us safe. Whether it is to measure the absorbency of tampons (21 CFR 801.430), enforce safety specifications of synthetic and natural wax coatings that are used to coat much of our produce, gummies and more (21 CFR 178.3770), quantifying the impurities in our bottled water (21 CFR 165.110) and many more.They have long been recognized as the de facto minimum standards that balance the need to protect consumers without imposing undue burdens on innovation by industry.